Safety of Corneal Collagen Cross linking With UV-A
Yakov Goldich, MD,* Arie L. Marcovich, MD,† Yaniv Barkana, MD,* Isaac Avni, MD,* and David Zadok, MD*
Purpose: To assess the possible damage to ocular tissues during treatment of keratoconus with UV-A–riboflavin corneal collagen cross-linking (CXL).
Design: Single center, prospective, interventional study.
Methods
Fourteen eyes of 14 patients aged 28.2 6 5.9 (mean 6 SD) years with progressive keratoconus were treated with UV-A– riboflavin CXL. Corneal endothelium was assessed with the endothelial specular microscope. Central retina was assessed with biomicroscopy fundus examination and with optical coherence tomography using macular thickness protocol. Patients were assessed preoperatively, at week 1, month 1, 3, 6, 9, and 12 after treatment.
Results: Comparative preoperative and postoperative results showed stable endothelial cell density (2730 cells/mm2, 2793 cells/mm2, and 2640 cells/mm2, preoperatively, at month 6, and at month 12, respectively) and stable foveal thickness (203, 202, and 205 mm, preoperatively, at month 6, and at month 12, respectively). No morphological abnormalities were noted.
Conclusions: UV-A–riboflavin CXL seems to be a safe procedure that does not cause damage to the corneal endothelium and central retina.
Key Words: keratoconus, corneal collagen cross-linking, UV-A (Cornea 2010;29:409–411)
Corneal collagen cross-linking (CXL) using UV-A light and riboflavin has been proposed recently as treatment to halt the progression of keratoconus (KC).1 Treatment effect
is reached by increasing covalent bindings between collagen molecules in the anterior corneal stroma. This process is mediated by free radicals generated by photosensitization of riboflavin by UV-A light.1,2 Both photochemically induced
Received for publication July 9, 2009; revision received August 9, 2009; accepted August 21, 2009.
From the *Department of Ophthalmology, Assaf Harofeh Medical Center, Zerifin, Israel; and †Department of Ophthalmology, Kaplan Medical Center, Rehovot, Israel.
No author has a financial or proprietary interest in any material or method mentioned.
Reprints: Yakov Goldich, MD, Department of Ophthalmology, Assaf Harofeh Medical Center, Beer Yaakov, Zerifin 70300, Israel (e-mail: doctor. goldich@gmail.com).Copyright 2010 by Lippincott Williams & Wilkins
free radicals and UV-A irradiation can potentially damage intraocular structures including corneal endothelial cells and human retina.3–6 Therefore, before we accept the UV-A– riboflavin CXL in everyday practice, we must ascertain its safety.
The aim of the current study was to assess in vivo the possible damage to the corneal endothelium and the retina after UV-A–riboflavin CXL treatment.
METHODS
Patients with KC were prospectively recruited from the cornea outpatient clinic of the Assaf Harofeh Medical Center. Inclusion criteria were progressive KC documented clinically within the past 12 months by astigmatic refraction and/or topography, age over 18 years, no previous ocular surgery, no corneal opacities, minimal central corneal thickness of 400 mm, and avoidance of contact lens wear for 1 month before initial evaluation and treatment. Patients were treated with UV-A–riboflavin CXL under aseptic conditions using topical preoperative anesthesia with oxybuprocaine hydro-chloride 0.4% drops (Localin; Fisher Pharmaceutical Labs). Treatment included 7-mm-diameter corneal deepithelializa-tion, instillation of 0.1% riboflavin in 20% dextran solution (Peschke Meditrade GmbH, Huenenberg, Switzerland) every 5 minutes for 40 minutes and corneal irradiation with UV-A 3 mW/cm2 (UV-X; Peschke Meditrade GmbH) for 30 minutes, 5 cm from the cornea. After the procedure, patients were treated with topical antibiotic (ofloxacin 0.3%, Oflox; Allergan) 4 times a day for 7 days, topical corticosteroid (dexamethasone 0.1%, Sterodex; Fisher Pharmaceutical Labs) 4 times a day for 1 month, and the eye was dressed with a soft therapeutic contact lens (Ocular Sciences, Ltd, Southampton, United Kingdom) for 3 days.
Patients were assessed preoperatively and at week 1, month 1, 3, 6, and 12 after treatment. Each examination included measurement of best spectacle–corrected visual acuity and slit-lamp examination and dilated fundus examina-tion. Corneal topography, pachymetry, endothelial cell density (ECD), central foveal thickness (CFT), and macular morphol-ogy (thickness and reflectivity) were assessed. Corneal topography and pachymetry were assessed preoperatively and at months 6, 9, and 12 with Orbscan II (Bausch & Lomb, Claremont, CA). ECD was assessed preoperatively and at months 1, 6, and 12 with the Konan Noncon Robo SP 6000 noncontact specular microscope (Konan Medical, Inc, Hyogo, Japan). CFT and macular morphology were assessed pre-operatively and at months 3, 6, 9, and 12 with Stratus optical
Cornea Volume 29, Number 4, April 2010 www.corneajrnl.com | 409
Goldich et al Cornea Volume 29, Number 4, April 2010
coherence tomography (OCT; Zeiss Humphrey Instruments, Dublin, CA).
The study was approved by the Institutional Ethics Committee of Assaf Harofeh Medical Center, and a written informed consent was obtained from each subject after the nature and intent of the study had been fully explained. The study protocol was consistent with the tenets of the Declaration of Helsinki.
Statistical Analysis
The data are presented as frequency or mean 6 SD. Paired 2-tailed Student t test was used to assess differences between the compared groups in ECD and CFT. The distributions of values within each data set were evaluated graphically. A P value of 0.05 was selected for the threshold of statistical significance. Analyses were performed using Excel (Microsoft, Corp, Redmond, WA).
RESULTS
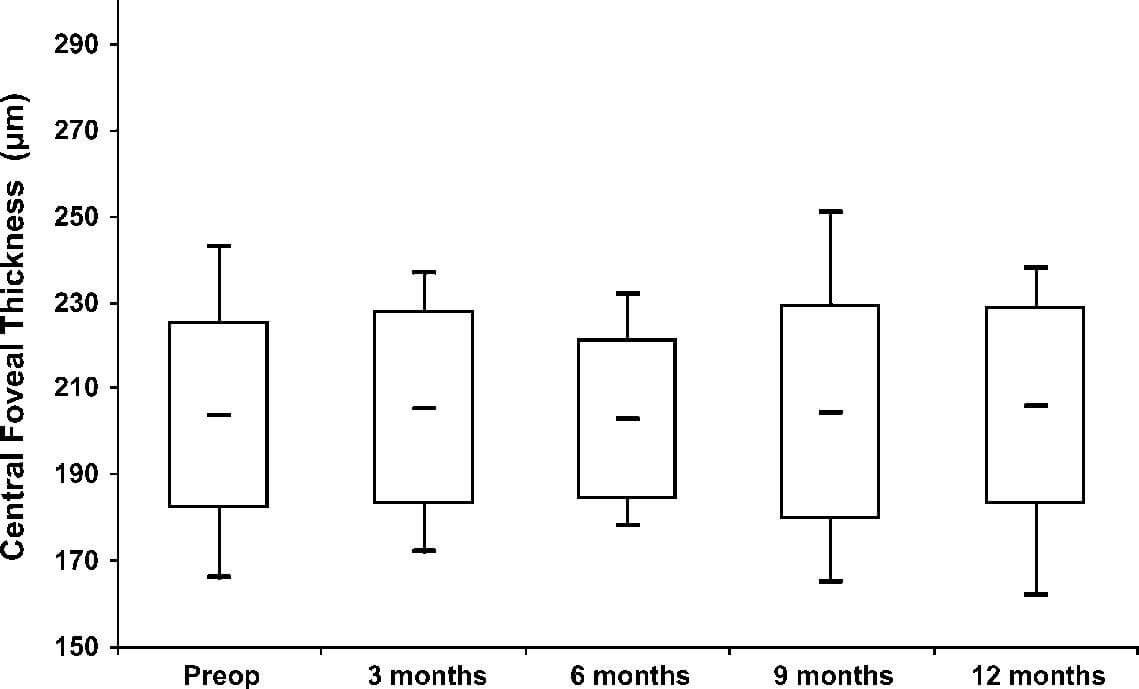
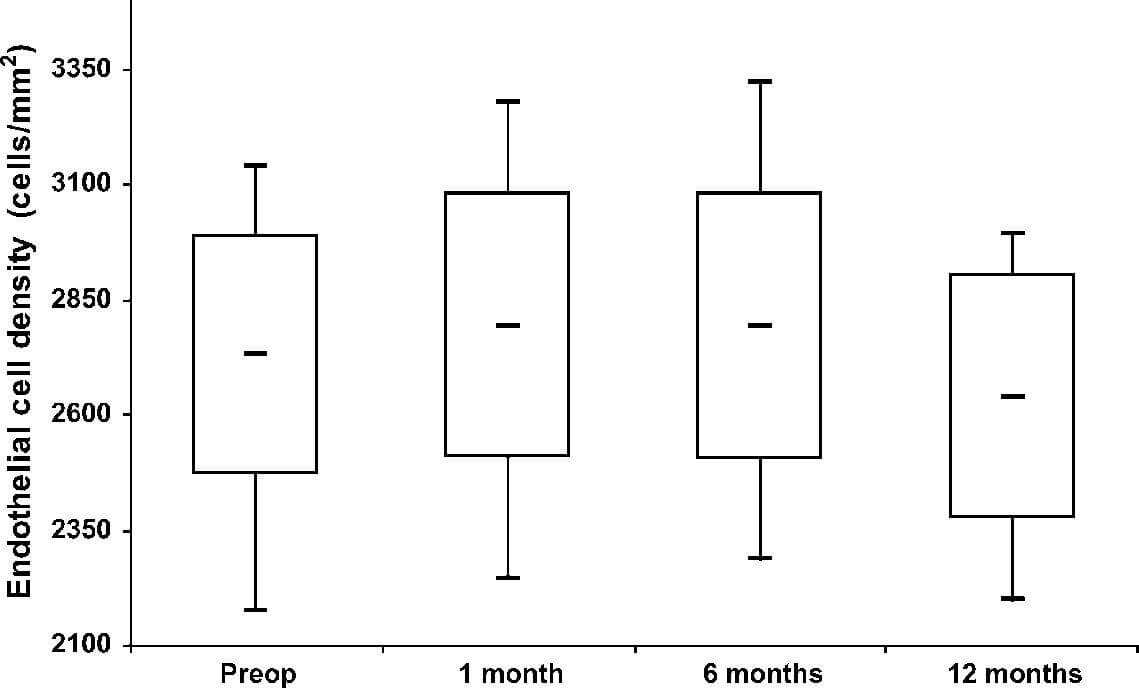
Fourteen eyes of 14 patients (8 males and 6 females) aged 28.2 6 5.9 years were included. Study parameters at baseline and 12 months postoperatively are shown in Table 1. Figures 1 and 2 present ECD and CFT measurements during the study period. There were no statistically significant differ-ences between preoperative and postoperative values of ECD and CFT at any follow-up time point.
No macular abnormalities were observed throughout the study period by means of biomicroscopic fundus examination and OCT analysis.
DISCUSSION
In the current study, we assessed the safety of treatment of KC using UV-A irradiation in conjunction with riboflavin. Our results show that this procedure does not cause corneal endothelial cell loss or has influence on foveal thickness. The recommended safety criteria, however, must be observed because UV irradiation has potential to damage various intraocular structures.4–6
FIGURE 1. Box (mean 6 SD) and whisker (smallest and largest values) plots showing ECD (cells/mm2) before treatment (Preop) and on 1 month, 6 months, and 12 months thereafter.
Based on in vitro studies, Wollensak et al1 recommended the following safety criteria for UV-A–riboflavin CXL: corneal thickness of at least 400 mm, irradiation with 3 mW/cm2 UV-A (370 nm), and keeping the cornea constantly covered with riboflavin solution before and during UV irradiation to provide a shielding effect.7 According to Lambert–Beer law, applying riboflavin increases UV-A absorption in the corneal stroma and reduces UV-A transmission through the cornea to about 7%. The endothelial damage threshold was shown to be at an irradiance of 0.35mW/cm2, which is approximately twice compared with the 0.18 mW/cm2 that reaches the corneal endothelium when using the currently recommended protocol.7,8
Previous human in vivo studies used confocal micros-copy9,10 and endothelial specular microscopy1,11 to assess endothelial damage. These studies did not detect any changes in endothelial morphology or density after UV-A–riboflavin CXL. However, different UV-A light sources were used in these studies. Caporossi et al9 and Mazzotta et al10 used CBM X-linker device (CSO, Florence, Italy). The CBM X-linker is.
TABLE 1. Study Parameters and Their Mean ChangeAfter Cross-linking
6 Months 12 Months Parameter Before CXL After CXL After CXL P BCVA (logMAR) 0.21 6 0.1 0.17 6 0.1 0.11 6 0.1 ,0.005UCVA (logMAR) 0.62 6 0.5 1.02 6 0.6 0.78 6 0.6 0.67CFT (mm) 203 6 21 203 6 17 205 6 22 0.78ECD (cells/mm2) 2730 6 261 2793 6 290 2640 6 266 0.21CCT (mm) 461 6 38 441 6 47 478 6 52 0.84Kmax (D) 53.9 6 5.9 53.1 6 5.5 52.1 6 5.0 0.006Kmin (D) 44.3 6 2.6 44.2 6 3.3 43.7 6 2.8 0.049Mean sim K (D) 46.2 6 2.8 46.3 6 3.3 45.6 6 2.9 0.14
Values are presented as mean 6 SD.
BCVA, best-corrected visual acuity with glasses; CCT, central corneal thickness; Kmax, maximal keratometry; Kmin, minimal keratometry; mean sim K, average simulated keratometry; P, t test (12 months after CXL versus before CXL); UCVA, uncorrected visual acuity.
410 | www.corneajrnl.com
FIGURE 2. Box (mean 6 SD) and whisker (smallest and largest values) plots showing CFT (mm) before treatment (Preop) and on 3, 6, 9, and 12 months thereafter.
q 2010 Lippincott Williams & Wilkins
Cornea Volume 29, Number 4, April 2010 Safety of UV-A–Riboflavin CXL
a system of 2 diodes, and the new system has 5 diodes that distribute UV light at a wavelength of no more than 370 nm. Wollensak et al1 used a light source with 2 UV diodes (Roithner Lasertechnik, Vienna, Austria) at a distance of 1 cm from the cornea. These original systems that used 2 lights could produce damaging hot spots. Wittig-Silva et al11 used UV-X system (Peschke Meditrade GmbH, Switzerland), which emits the UV light via a special optical system (Koehler optics) that makes the unit much less responsive to slight variations in illumination distance. This way, the risk of overexposure is minimized. In the UV-X system, 7 light emitting diodes are set in a flower pattern. This gives a very smooth distribution of light over the cornea. In our study, we used only the UV-X illumination source with 7 diodes, and our results show no significant decline in ECD at any time after UV-A–riboflavin treatment with follow-up of 1 year (P = 0.21). Similar stability of ECD was reported by Wittig-Silva et al11 using similar irradiation system.
Another important aspect is the safety of UV-A– riboflavin treatment to inner eye structures, especially to the lens and the retina.3,5,6 Light injury to the retina may be caused by thermal- or light-induced photochemical damage. Ultravi-olet radiation is potentially more hazardous to the retina than other wavelengths.12 In rabbits, the threshold radiant exposure for retinal damage was shown to be 4.3 mW/cm2,5,6 which is higher than dose during CXL (3 mW/cm2). In the human eye, the main absorption of the UV-A light is in the crystalline lens and only a small fraction may reach the retina.5,6 However, a clear crystalline lens that is characteristic of young patients with KC may be less protecting against such UV penetration. Although in vitro studies showed that the shielding effect of riboflavin limited radiant transmission to inner ocular struc-tures,13 no previous in vivo studies were conducted to assess retinal morphological changes after UV-A–riboflavin CXL.
Both clinical biomicroscopy fundus examination and OCT may reveal retinal photochemical damage.14–17 Retinal photochemical damage has been described as solar retinopathy in patients with a history of sun gazing. Considering that over 90% of the sun’s UV light reaching earth is UV-A light (320– 400 nm),18 we may expect that artificially induced damage from UV-A irradiation during CXL (370 nm) will have similar morphological characteristics to solar retinopathy. OCT was previously shown to be a useful tool in assessing retinal photochemical damage. Various patterns of macular damage after prolonged exposure to solar radiation were described. Garg et al,17 Stangos et al,16 and Gulkilik et al15 described OCT findings in patients with chronic solar retinopathy. They all reported hyporeflective space at the level of the outer neurosensory retina and retinal pigment epithelium with foveal thinning. Bechmann et al14 described OCT findings in patients with early solar retinopathy and reported a hyperreflective area involving all foveolar retinal layers with normal foveal thickness. In our study, all OCT examinations throughout the study period of 1 year presented a normal macula in every studied eye. No changes in macular thickness or in retinal reflectivity in any layer were observed. Clinical funduscopic examinations were unremarkable during every time point, and no morphological changes were noted. Grewal et al19 recently published 1-year follow-up results after CXL in progressive
q 2010 Lippincott Williams & Wilkins
KC. Using Stratus OCT, they reported a slight decrease of 29 mm in foveal thickness 1 year after cross-linking. Although this change was not statistically significant, its magnitude is different from the practically stable foveal thickness as mea-sured in our study. This suggests that more studies with larger sample size and longer follow-up are required to understand whether this difference is meaningful.
In addition, in the current study, we assessed safety using anatomic parameters. Further studies should be done to assess safety using functional testing, such as electroretinography.
In conclusion, our data suggest that UV-A–riboflavin CXL is a safe procedure for treating progressive KC. When this procedure is performed using currently recommended parameters, no corneal endothelium or retinal structural damage is expected.
REFERENCES
1. Wollensak G, Spoerl E, Seiler T. Riboflavin/ultraviolet-a-induced collagen crosslinking for the treatment of keratoconus. Am J Ophthalmol. 2003; 135:620–627.
2. Kohlhaas M, Spoerl E, Schilde T, et al. Biomechanical evidence of the distribution of cross-links in corneas treated with riboflavin and ultraviolet A light. J Cataract Refract Surg. 2006;32:279–283.
3. Dovrat A, Weinreb O. Effects of UV-A radiation on lens epithelial NaK-ATPase in organ culture. Invest Ophthalmol Vis Sci. 1999;40:1616–1620.
4. Ambach W, Blumthaler M, Schopf T, et al. Spectral transmission of the optical media of the human eye with respect to keratitis and cataract formation. Doc Ophthalmol. 1994;88:165–173.
5. Zigman S. Environmental near-UV radiation and cataracts. Optom Vis Sci. 1995;72:899–901.
6. Pitts DG. Glenn A. Fry Award Lecture—1977. The ocular effects of ultraviolet radiation. Am J Optom Physiol Opt. 1978;55:19–35.
7. Wollensak G, Sporl E, Reber F, et al. Corneal endothelial cytotoxicity of riboflavin/UVA treatment in vitro. Ophthalmic Res. 2003;35:324–328.
8. Wollensak G, Spoerl E, Wilsch M, et al. Endothelial cell damage after riboflavin-ultraviolet-A treatment in the rabbit. J Cataract Refract Surg. 2003;29:1786–1790.
9. Caporossi A, Baiocchi S, Mazzotta C, et al. Parasurgical therapy for keratoconus by riboflavin-ultraviolet type A rays induced cross-linking ofcorneal collagen: preliminary refractive results in an Italian study.J Cataract Refract Surg. 2006;32:837–845.
10. Mazzotta C, Balestrazzi A, Traversi C, et al. Treatment of progressive keratoconus by riboflavin-UVA-induced cross-linking of corneal colla-gen: ultrastructural analysis by Heidelberg retinal tomograph II in vivo confocal microscopy in humans. Cornea. 2007;26:390–397.
11. Wittig-Silva C, Whiting M, Lamoureux E, et al. A randomized controlled trial of corneal collagen cross-linking in progressive keratoconus: preliminary results. J Refract Surg. 2008;24:S720–S725.
12. Ham WT Jr, Mueller HA, Sliney DH. Retinal sensitivity to damage from short wavelength light. Nature. 1976;260:153–155.
13. Spoerl E, Mrochen M, Sliney D, et al. Safety of UVA-riboflavin cross-linking of the cornea. Cornea. 2007;26:385–389.
14. Bechmann M, Ehrt O, Thiel MJ, et al. Optical coherence tomography findings in early solar retinopathy. Br J Ophthalmol. 2000;84:547–548.
15. Gulkilik G, Taskapili M, Kocabora S, et al. Association between visual acuity loss and optical coherence tomography findings in patients with late solar retinopathy. Retina. 2009;29:257–261.
16. Stangos AN, Petropoulos IK, Pournaras JA, et al. Optical coherence tomography and multifocal electroretinogram findings in chronic solar retinopathy. Am J Ophthalmol. 2007;144:131–134.
17. Garg SJ, Martidis A, Nelson ML, et al. Optical coherence tomography of chronic solar retinopathy. Am J Ophthalmol. 2004;137:351–354.
18. ASTM. Terrestrial Global Spectral Irradiance Tables for Air Mass 1.5. Document 138 RI E 44.02; 1981.
19. Grewal DS, Brar GS, Jain R, et al. Corneal collagen crosslinking using riboflavin and ultraviolet-A light for keratoconus: one-year analysis using Scheimpflug imaging. J Cataract Refract Surg. 2009;35:425–432.